- About
- Expertise
- Services
- Testing Services Overview
- Research Models
- GLP Studies & FDA Assistance
- Alprazolam (Xanax)
- Armodafinil (Nuvigil)
- Carisoprodol (Soma)
- Clonazepam (Klonopin)
- Ivermectin (Stromectol)
- Lorazepam (Ativan)
- Modafinil (Provigil)
- Molnupiravir (Lagevrio)
- Prednisone (Deltasone)
- Pregabalin (Lyrica)
- Sumatriptan (Imitrex)
- Testosterone (Cernos)
- Zolpidem (Ambien)
- Zopiclone (Imovane)
- Support
- News/Events
- Blog
- Contact
Expertise
Your Trusted Pharmaceutical, Biotech, and Medical Device CRO with Proven Preclinical Expertise
The Surpass team, in the greater Minneapolis/Twin Cities, has hundreds of years of collective preclinical study management and execution experience across numerous medical disciplines. We support you every step of the way, from study design through execution and reporting.
At Surpass our innovative approach to preclinical research goes beyond the ordinary; we strive to be more than a typical preclinical CRO partner. Our goal is to be a valuable member of your product development team, helping you design the most translatable study, the first time. From Cardiovascular to Wound Healing/Traumatic Injury to Respiratory studies, and much more, Surpass has experience and knowledge to help you advance your new device or therapy to market. Whether you are testing a de novo or disruptive technology or need to evaluate an iterative medical device design, pharmaceutical or biologic, Surpass will partner with you to make your project successful. Contact Surpass to discuss your upcoming project with our veteran Research Team.
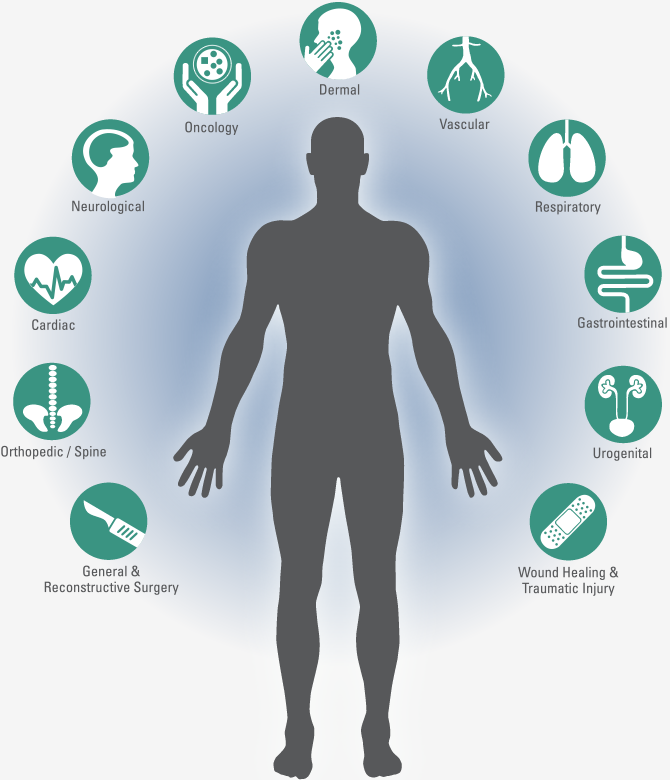
Do You Need A Preclinical CRO You Can Count On?
Surpass can help. Whether you need a specialized Medical Device CRO or a pharmaceutical or biologic testing partner, the Surpass team has experience conducting preclinical in vivo and human cadaveric studies with test articles across the following therapeutic disciplines:
- Cardiology - Interventional and Electrophysiology (EP)
- Colon and Rectal Surgery
- Dermatology
- Emergency Medicine
- Endocrinology, Diabetes and Metabolism
- Gastroenterology
- Interventional Radiology
- Neurology
- Obstetrics and Gynecology
- Oncology
- Ophthalmology
- Orthopaedic Surgery
- Otolaryngology (ENT)
- Plastic and Reconstructive Surgery
- Pulmonary Disease
- Regenerative Medicine
- Sleep Medicine
- Thoracic and Cardiac Surgery
- Tissue Engineering
- Urology
- Vascular Neurology
- Vascular Surgery
- And more
Medical Devices, Pharmaceuticals, Biotech Experience
Do you need a preclinical CRO with proven expertise to test your innovative medical device or therapy?
Surpass can help. With more than 500 different client companies served through the years, Surpass has experience testing a variety of different medical devices, pharmaceuticals, and biologics at our Minneapolis/Twin Cities location.
Medical Devices and Therapies:
- Ablation systems (radiofrequency (RF), microwave, cryo, ultrasonic)
- Aneurysm grafts, coils, bridging stents, fillers, and flow diverters for cerebral and peripheral applications
- Antiepileptics
- Anticoagulation agents
- Atrial fibrillation (Afib) and Atrial flutter treatments (pulmonary vein ablation and left atrial (LA) appendage occlusion)
- Bariatric implants
- Bioabsorbable materials
- Bone anchors
- Bone cement
- Bone marrow extraction devices
- Bowel anastomotic devices
- Brachytherapy
- Cancer treatments (breast, cervix, liver, kidney, pancreas, prostate)
- Chemoembolization
- Colorectal/gastrointestinal/stomach stapling systems
- Combination drug/device, cell/device products
- COVID-19 drugs
- Debridement solutions and therapies
- Defibrillators
- Drug pumps
- Dialysis treatments
- Dressing materials and methods
- Drug coated/eluting balloons
- Electrical stimulators
- Electrosurgical devices
- Electrophysiology mapping systems and telemetry monitors
- Embolic protection devices
- Endotracheal tubes
- Gastrointestinal implants
- Gene and Cell Therapies
- Heart valves
- Hemostatic and coagulation devices/agents
- Hormone replacement therapy
- Imaging systems
- Implantable leads
- Implantable pumps
- Incontinence treatments
- Infectious disease treatments
- Infusion pumps
- Lasers
- Neuromodulators and stimulators
- Ocular imaging systems (retinal/corneal)
- Orthopedic implants
- Pacemakers
- Pharmaceutical agents
- Polymer materials
- Pulmonary sealants
- Pulmonary stents
- Reconstructive surgical implants
- Renal denervation devices
- Retractor systems
- Spinal fusion devices
- Stents (bioabsorbable, drug eluting, covered, bifurcating, coronary, neurological, endovascular, and peripheral)
- Telemetry systems
- Therapeutic hyperthermia/hypothermia induction devices
- Tendon repair devices
- Tissue sealants
- Ureteral stent
- Urethral balloons
- Urethral debulking
- Vascular closures
- Vena cava filters
- Venous insufficiency devices (valves/reflux)
- Wound closures
- And more
Surpass is uniquely qualified as your Medical Device CRO and pharmaceutical/biologics preclinical testing partner to make your projects successful. Contact Surpass today.
We're With You
Every Step of the Way.